Forschung
Polymere - Kettenmoleküle – sind außerordentlich vielseitige Materialien, die wir aus vielen Anwendungen im Alltag wegen ihrer besonderen mechanischen, optischen oder elektrischen Eigenschaften kennen. Auch biologische Materie besteht zu einem Großteil aus Polymeren. Polymere besitzen eine ganze Reihe typischer, besonderer Eigenschaften, wie zum Beispiel Viskoelastizität, Kautschukelastizität, Teilkristallinität oder Selbstorganisation.
Welche Forschungsthemen bearbeiten wir?
1. Grenzflächeninduzierte Kristallisation
https://pubs.acs.org/doi/10.1021/acs.jpclett.9b00608
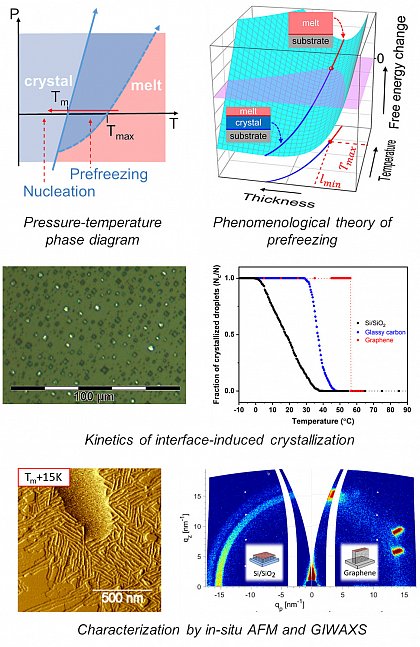
Die Initiierung der Polymerkristallisation an einer Grenzfläche zu einem Festkörper kann die teilkristalline Morphologie und die Kristallisationskinetik stark beeinflussen, die in der Regel die der homogenen Keimbildung im Volumen übertrifft. Im Allgemeinen kann eine feste Oberfläche die Kristallisation entweder durch heterogene Keimbildung oder durch Prefreezing induzieren. Während es sich bei der heterogenen Keimbildung um einen aktivierten Prozess handelt, der unterhalb des Schmelzpunkts Tm stattfindet, entsteht beim Prefreezing eine dünne kristalline Schicht oberhalb von Tm, die sich beim Abkühlen auf Tm verdickt. Sowohl das Prefreezing als auch die heterogene Keimbildung sind von den Grenzflächenwechselwirkungen zwischen einem Polymer und dem darunter liegenden Substrat abhängig. Durch die Kombination experimenteller und theoretischer Ansätze ist es unser Ziel, die Materialparameter zu verstehen, die die Thermodynamik und Kinetik der grenzflächeninduzierten Polymerkristallisation beeinflussen.
Zu diesem Zweck untersuchen wir die Kristallisation von dünnen Filmen und isolierten Tröpfchen aus teilkristallinen Polymeren auf festen Substraten. Als experimentelle Methoden verwenden wir die optische Mikroskopie mit polarisiertem Licht (POM), die Rasterkraftmikroskopie (AFM) und die Röntgenweitwinkelstreuung unter streifendem Einfall (GIWAXS).
2. Kristallisation von biologisch abbaubaren Polymeren
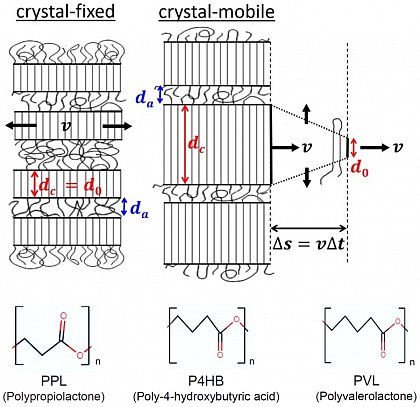
Beispiele von Polyestern mit unterschiedlicher Anzahl von Methylengruppen (CH2)
Biologisch abbaubare Polymere sollen Standardpolymere in verschiedenen
Anwendungen ersetzen. Bisher sind aber z.B. die mechanischen
Eigenschaften der biologisch abbaubaren Alternativen noch nicht gut
genug für den breiten Einsatz. Das liegt z.T. an der polymerspezifischen
teilkristallinen Struktur.
Polymere mit regelmäßigem chemischen Aufbau kristallisieren in einer
teilkristallinen Struktur. Es bilden sich kristalline Lamellen mit
ungeordneten/amorphen Zwischenschichten. Diese haben Dicken im
Nanometerbereich und sind lateral bis zu einigen Mikrometern groß.
Die entstehende Nanostruktur beeinflusst maßgeblich die makroskopischen
Materialeigenschaften und ergibt sich sowohl aus dem chemischen Aufbau
der Polymere als auch aus deren Verarbeitung. Die chemische Struktur
bestimmt neben der Anordnung der Ketten auch deren Beweglichkeit in den
kristallinen Lamellen. So lassen sich Polymere anhand ihrer
Beweglichkeit in crystal-mobile und crystal-fixed einteilen. Die
unterschiedliche Beweglichkeit führt zu großen Unterschieden in der
teilkristallinen Nanostruktur während und nach der Kristallisation.
Wir untersuchen den Zusammenhang zwischen der teilkristallinen Struktur
und den makroskopischen Eigenschaften anhand von biologisch abbaubaren
Polyestern mit verschieden langen Methylensequenzen mit und ohne
Seitenketten. Die Dynamik der Ketten untersuchen wir in der Schmelze mit
Rheologie und im teilkristallinen Zustand mit NMR. Die sich ergebende
Nanostruktur wird mit Röntgenkleinwinkelstreuung vermessen. Für deren
Auswertung entwickeln und programmieren wir Analysewerkzeuge.
3. Strukturbildung in Halbleitenden Polymeren
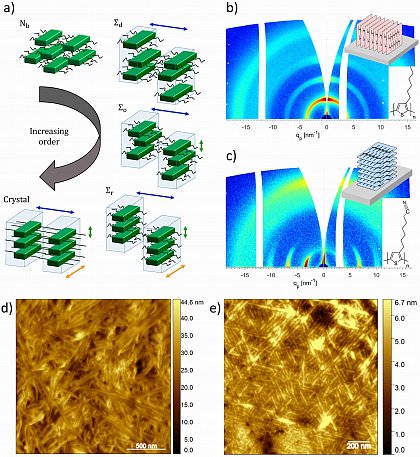
a) Verschiedene Ordnungszustände von flüssigkristallin bis kristallin, die sich in halbleitenden Polymeren bilden können (https://doi.org/10.1021/acs.jpcb.1c10599).
b,c) Streuungsbilder von P3HT auf Silizium (b) mit Edge-on-Orientierung und P3CNHT auf Graphen (c) mit Face-on-Orientierung. Die chemischen Strukturen des Monomers und die entsprechenden Kristallorientierungen sind als Insets dargestellt.
d,e) Höhenaufnahmen mit dem Rasterkraftmikroskop von dünnen Filmen von P3HT auf Siliziumsubstrat (d) und P3CNHT auf Graphen-Substrat (e).
Halbleitende Polymere sind vielversprechende Materialien für ein breites Spektrum organischer elektronischer Anwendungen wie Solarzellen, Transistoren und Sensoren. Da die Ladungstransporteigenschaften in teilkristallinen halbleitenden Polymeren anisotrop sind, spielen die teilkristalline Morphologie und ihre Ausrichtung in dünnen Filmen eine wichtige Rolle für die Effizienz organischer elektronischer Bauelemente. Wir untersuchen die Strukturbildung in halbleitenden Polymeren in Volumen und in dünnen Filmen unter dem Einfluss der molekularen Zusammensetzung und Architektur, des Molekulargewichts und vor allem der Grenzflächen. Unsere Ergebnisse zeigen, dass die Grenzflächen zu den Substraten und zum Vakuum den Kristallisationsmechanismus stark beeinflussen, der wiederum die Kristallorientierung bestimmt, die in Filmen aus halbleitenden Polymeren gebildet wird. Unser Ziel ist es, diese grenzflächeninduzierten Ordnungsphänomene besser zu verstehen und die Kristallorientierung in Filmen aus halbleitenden Polymeren durch Veränderung der Grenzflächenwechselwirkungen zu steuern.
Um diese Ziele zu erreichen, wird die teilkristalline Morphologie in Bulk-Proben und dünnen Schichten bei Raumtemperatur und erhöhten Temperaturen mit verschiedenen Röntgenstreutechniken und Rasterkraftmikroskopie untersucht, während Grenzflächenwechselwirkungen mit Hilfe von Kontaktwinkelmessungen analysiert werden.
4. Mechanische Eigenschaften von teilkristallinen Polymeren
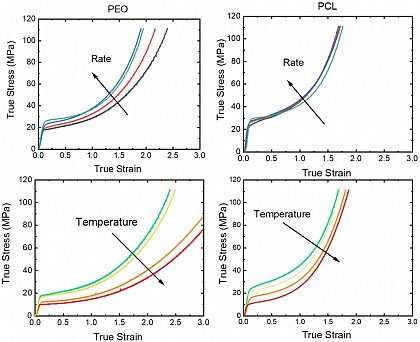
Mechanische Eigenschaften von teilkristallinen Polymeren
Die mechanischen Eigenschaften von Polymeren sind eine komplexe multivariable Funktion von intrinsischen und extrinsischen Faktoren. Einer der intrinsischen Faktoren, die sie beeinflussen, ist die Kristallisation; es ist bekannt, dass sich die Eigenschaften von teilkristallinen Polymeren stark von amorphen unterscheiden. Um die Auswirkung der Kristallinität auf die Polymere festzustellen, wird in der Regel Polyethylen oder Polypropylen verwendet. Eine Diskussion über das universelle Verhalten von teilkristallinen Polymeren ist jedoch notwendig, um neue Materialien zu erforschen, die den Umweltproblemen und den Anwendungsanforderungen gerecht werden. Die Kettenmobilität ist einer der kritischen Aspekte vieler Eigenschaften von Polymermaterialien, einschließlich der mechanischen Leistung; die Diskussion über die Kettenmobilität könnte über die amorphe Phase hinaus auf den Kristall ausgedehnt werden; in diesem Fall könnte der Einfluss auf die interkristalline Kettendiffusion (intercrystalline chain diffusion - ICD), die die kristallinen Segmente in verschiedenen Zeitskalen ausdrücken können, untersucht werden. Unsere Forschung konzentriert sich auf das Verständnis des Einflusses der Parameter, die die kristalline Morphologie und die Kristallnatur auf die mechanischen Eigenschaften beeinflussen.